Dragon Fire
Dr Mendil strode before the batch of new dragon handlers. “All right, folks!” He called out. “Quiz for you all: how hot can a dragon’s breath get?”
A hand shot up from an eager newbie. “The last time they measured it, wasn’t it 1000 degrees?”
Dr. Mendil held up his arms in a big X. “Nope, thanks for playing. The answer is: hot enough to burn your faces off! And that’s why,” He held up a body suit made of the thickest leather they could stitch together. “You’ll be wearing one of these bad boys every time you go near one of our juveniles. As we say in the business, ‘living is quite fashionable.’ Time to try them on for a test run.”
Once they were all properly suited up, Dr. Mendil led his new team down a deserted hallway. Deserted, save for the juvenile dragons nipping at the bars of their roosting nests. “We usually let our dragons free roam,” Dr. Mendil said. “But our feistier dragons need to be fed before we let them out. You’ll see why.” He flipped down his suit’s black visor. “I have in my hand a nice, wet steak. Now, our dragons shouldn’t be territorial around food, but…” Dr. Mendil waddled towards one of the nests and held out the slab of meat. The dragon within lunged forward and tried to shove her mouth between the bars to snatch the detectible treat. “We have a select few who act like they’re constantly starving. Teenagers, am I right? Even across species, they can be—”
A torrent flames struck Dr. Mendil, completely coating him in red and white light. One of the new handlers shrieked. Another fainted. Once the flames died down, Dr. Mendil stood tall in his smoking fire suit. The scent of singed leather filled the hall. Dr. Mendil tossed the now-charred steak through the bars and lifted his visor. “….Pushy,” he finished.
What do you think of when you hear “dragons”?
Dragon Fire.
Name a dragon story, and it probably has a dragon spewing fire (there are the occasional alternate dragon breathes, like ice or acid, but those are few and far between). Smaug from The Hobbit? Breathes fire. Fafinir from Norse Mythology? Usually depicted as breathing fire. All the dragons from Game of Thrones (or A Song of Ice and Fire)? All fire spitters.
Spewing or spitting as a self-defense is not uncommon in the animal kingdom. To name a few such species that hold some impressive spitting records: spitting cobra, camels, spitting spiders, and the archerfish. So, our fantasy dragon is in good company (evolutionarily).
Let’s roll up our sleeves and science this dragon breath!
Previous Attempts at Sciencifying Dragon Fire

When I’ve posed the question, “how would dragon fire work” to my other scientist friends, many have told me to check out “A Flight of Dragons,” a story that apparently has already answered this post’s very topic.
Peter Dickinson’s “A Flight of Dragons” explains a dragon’s fiery breath and flight through a clever bit of fantasy evolution: dragons swallow gemstones and limestone that combine into their “craw.” A craw is kind of like a place birds and insects can store food before it gets digested, and in Peter Dickinson’s world, dragons use this organ too. The gems grind up the limestone in the specialized organ, releasing the limestone’s calcium. This then mixes with their stomach acid, producing hydrogen gas.
For those who’ve taken chemistry in the past, this is a classic oxidation-reduction reaction: Ca + 2HCl –> CaCl + H2. Calcium is oxidized, and hydrogen is reduced.
For those who’re a little out of chemistry practice, all you need to know is that calcium plus acid leads to salt and hydrogen gas.
In this fantasy world, the dragons have “compartments” in their body that they can expand or contract to change the density of the stored hydrogen, allowing them to float/fly. When they breathe out fire, they’re expelling this very same hydrogen out of their mouth. A small organ at the top of their mouth produces a spark (akin to the electricity produced biologically by an electric eel) that ignites their breath weapon.
Presto! We have our fire belch!
Except…Do we? This is a fine explanation, but there are some glaring issues:
1) The amount of gas necessary to lift a dragon seems like…a lot. I performed a few back-of-the-envelope calculations. Let’s assume that a dragon weighs as much as an elephant, which I found to be 13,200 lbs (on the heavier side of the possible range). I then converted that to Newtons (58716.525321N) and then used this calculator to calculated the volume of hydrogen needed to lift that force. I got 4981.2140 m^3. That’s 5,000,000 liters. Look at your biggest water bottle (that’s about a liter) and imagine 5,000,000 of those. That’s twice the volume of an Olympic swimming pool and that’s not even considering the rest of the dragon, like the lungs and wings and everything!
2) Hydrogen-produced flames are really hard to see as it’s a gentle light blue. During the day, your naked eye will probably not be able to see it.
For number 1, many readers go, “yeah, but they can store that much hydrogen in them because magic,” and that’s fair. As I’ve described in my other blog posts, flight in creatures bigger than a bird is near impossible due to physics and biology. Flying dragons could only exist by magically breaking a few of nature’s laws, so why should we be so picky about where a tiny bit of hydrogen is stored?
I think number 2 is by far the most egregious of the issues. Igniting a barely visible flame? Where’s the theatricality?! The cinema potential?! While producing a scarely-visible flame would be beneficial to the dragon for hunting, that’s not why we read fantasy books. I want my retinas to be burned with amazement, dang it!
We can do better.
A Discussion on Oil
After diving into the lore of Peter Dickinson’s world, I then checked the internet for explanations on plausible dragon breath, this time from modern science communicators. The YouTube channels The Nerdist and How Stuff Works have done videos exploring.
One possible breath weapon mentioned in a video was methane, though methane has similar issues as hydrogen (specifically the “where could that much gas be stored in a creature” issue). Again, storage is an inherent problem when you use gas as the fire’s fuel (even if we compressed that volume, the space the gas would take up would still be impressive). Ideally, we have a fuel source that doesn’t require a ton of space in our dragon.
I did check out whether we could use oil as the fuel, but it turns out a lot of naturally occurring oil isn’t easily ignitable. Like, it’s impressively hard to make certain oils catch fire. This is because it takes a lot of energy to catch oil on fire (though once it catches fire, it burns strong, and it burns long). You’ve heard of this called “flashpoint,” or the temperature that an oil needs to be at before it ignites. And this is a good thing about oil; fats in general should be harder to “burn,” which is why animal bodies use fat as energy storage.
The problem with needing to add a ton of energy to ignite a dragon’s breath is that it starts to be evolutionarily unfavorable. Say you’re a dragon; it’s not great to have your breath weapon take up so much of your energy to ignite. Whatever mechanism you use to ignite the oil will tire you out because you must sacrifice your energy to burst that oil into flame. This is the constant question in evolution: maximizing energy use. At what point does it become so energetically unfavorable to use oil for dragon breathe? What if it tires you out so much that after a few breathes, you’re too tired to fly or to fight (if you, the dragon, are in a battle)?
(This explains why so many scientists look at flammable gases for plausible dragon fire. It doesn’t take a ton of energy to burst all that gas into flame).
I feel like we can do better than oil.
There’s discussion how dragon fire could work similarly to the bombardier beetle. The beetle has two reservoirs that contain hydroquinones and hydrogen peroxide. You’ve probably heard of hydrogen peroxide—it works great on blood stains—but what the heck are hydroquinones? They are small molecules that can be used to develop photos. When the beetle releases its “contents” in short, fast bursts (kind of like a gatling gun), the compounds mix to make a hot liquid mixture that the beetle shoots out of its rear end.
And when I say hot, I mean 100 degrees, water-boiling hot.
Why is that? Well, it turns out that when the beetle dumps the hydroquinones and hydrogen peroxide from the reservoirs into the exit chamber, the chamber is lined with enxymes that break down both components into quinones and water. This is a highly exothermic (or “heat forming”) reaction, releasing 48.5 kcal/mole in a reaction. You’ll have to trust me on this when I say that’s a ton of energy made for such a tiny reaction. Within a fraction of a second, the bombardier beetle can shoot your hand with scalding hot liquid. It’s so hot, it’s estimated that 1/5 of the mixed contents get evaporated when it’s made in that exit chamber.
The obvious problem with this mechanism for our dragon: while this is hot …It ain’t fire.
We want FLAMES. No respectful fantasy town is going to be leveled by boiling hot water spray.
But maybe we can use the beetle’s biological reservoirs for our dragon? What if we put those glands in the neck of our dragon and have the
What if they’re just shooting out water?
Actually, let’s steal from the bombardier beetle and shoot out TWO things: water and some metal-ladened oil. And don’t worry: the oil isn’t what we’re igniting.
Setting Metals Ablaze
Did you know that there are metals that ignite in water and oxygen?
Take lithium, for example. Or sodium. These are alkali metals, which inhabit the whole first column on the periodic table (see below). These metals are extremely reactive, and become increasingly reactive as you go down the column. Check out this video here to see examples of the explosions these metals make (my favorite is the cesium one!).
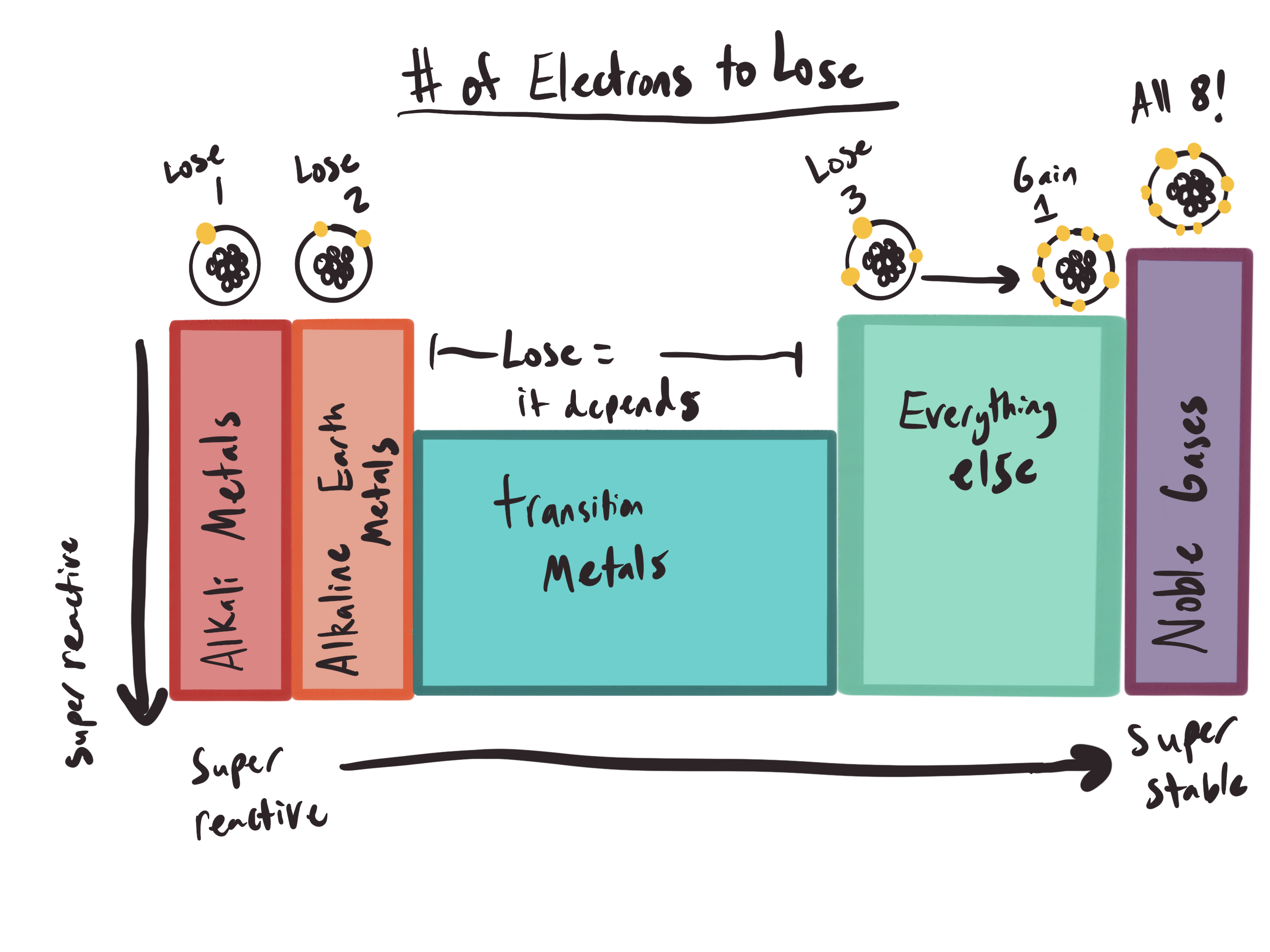
Why are these metals so reactive? Well, if you look at the image I so expertly created in procreate, you’ll see that I marked how many electrons are on the outside of a given atom over each corresponding column (I kind of skipped the transition metals because they’re weird and not applicable to this post). Now, every atom is most stable when they have 8 electrons in their outermost shell. This is called a “stable octet.”
All the way to the right of the periodic table, the noble gases have the stable octet and are, as their name suggests, extremely stable. One column to the left of the noble gases, you have atoms with 7 electrons. You can technically try to rip away those 7 electrons to make a stable octet, but it’s more efficient to just add an electron to the atom to make the full octet. Once you get to below 4 electrons, then it becomes favorable to remove the electrons to get to the stable octet. Alakline metals have 2 electrons, so you’ll need to rip away the
Alaki metals only have to lose 1 electron.
One. Tiny. Electron.
That means it’s very, very easy to remove that single electron. This equals “high reactivity,” which equals “more explosive in water.” This high reactivity increases even more as we move down on the column (this has to do with the atomic radius, but that’s getting too deep into the chemistry weeds).
When we drop these highly reactive metals (with a very loose single electron) into water, the electron flies off, leaving behind a positive, unreactive alkali metal atom. This generates a truckload of heat. That electron then interacts with water to produce oxygen and, importantly, hydrogen. The heat produced from the electron jumping from the alkali emtal atom ignites hydrogen, producing an impressive flame.
Ironically, we do ultimately have hydrogen gas as our fire fuel, just not in the way we expected.
It turns out this reaction is even more complicated than chemists realized. In 2015, this paper by Mason et al examined
This sudden change into positively charged atoms causes what is called a “coulomb explosion,” which where the charged atoms are repelled away from the metal, leading to a cyclical effect where even more alkali metal atoms are exposed to water, leading to the loss of electrons and the production of hydrogen, leading to even more metal atoms exposed to water, and you get the idea.
Could we use alkaline metals (second column of the periodic table)? Not really. They don’t react quite as intensely as the alkali metals, bubbling. It’s still impressive for a chemistry demonstration in front of children, but it wouldn’t provide the flames necessary for our dragon.
What if—hear me out—whatever substance the dragon shoots out is equivalent to mineral oil? I’ve worked with reactive alkali metals in the lab back in the ol’ organic chemistry days, and we had to store the soft metals in mineral oil to keep it from reacting with the oxygen in the air. It makes sense that the dragons’ storage reservoirs in their neck would produce an oil or fat that keeps their alkali metals from reacting preemptively.
Where would dragons get those pure alkali metals? It’s tempting to say from eating metals from their gold hoard, honestly. Their metabolism could isolate these metals much like our body can isolate iron for our red blood cells. In the wild, dragons could also be near areas that are also prime mining sites (hence why they’d take up residence in dwarven kingdoms). If humans, or any other humanoids, shove the dragons out of their habitats and dig up their valuable metal, then the dragons adapt and take the metal back from humans in the form of coin, swords, and any other shiny material.
Except for one eensy teensy problem…Pure alkali metals aren’t really found in nature, nor in the treasures that we humans make. That’s because they’re so reactive. Alkali metals are found in salts, because, well, they’ve already reacted. To get the reactive forms of the metals, we have to fun electricity through salts at extremely high temperatures (see the Castner Process). There’s no way a dragon would be able to do this at the cellular level.
Hmm… Perhaps this is a biological mystery that fantasy scientists are still figuring out? Maybe dragons eat lots of salts and they have a special metabolism and biological process that helps them produce the reactive metals? Shrugs “Because magic?”
(Look, the hydrogen explanation has storing the hydrogen produced by limestone and stomach acid somewhere in their body and requires the dragon to have an “ignition organ” at the top of their mouth to ignite the hydrogen. I’d argue my metal-metabolism is as wild as that!)
Scientifically Accurate Dragon Fire According to Yours Truly
Here is the mechanism I propose:
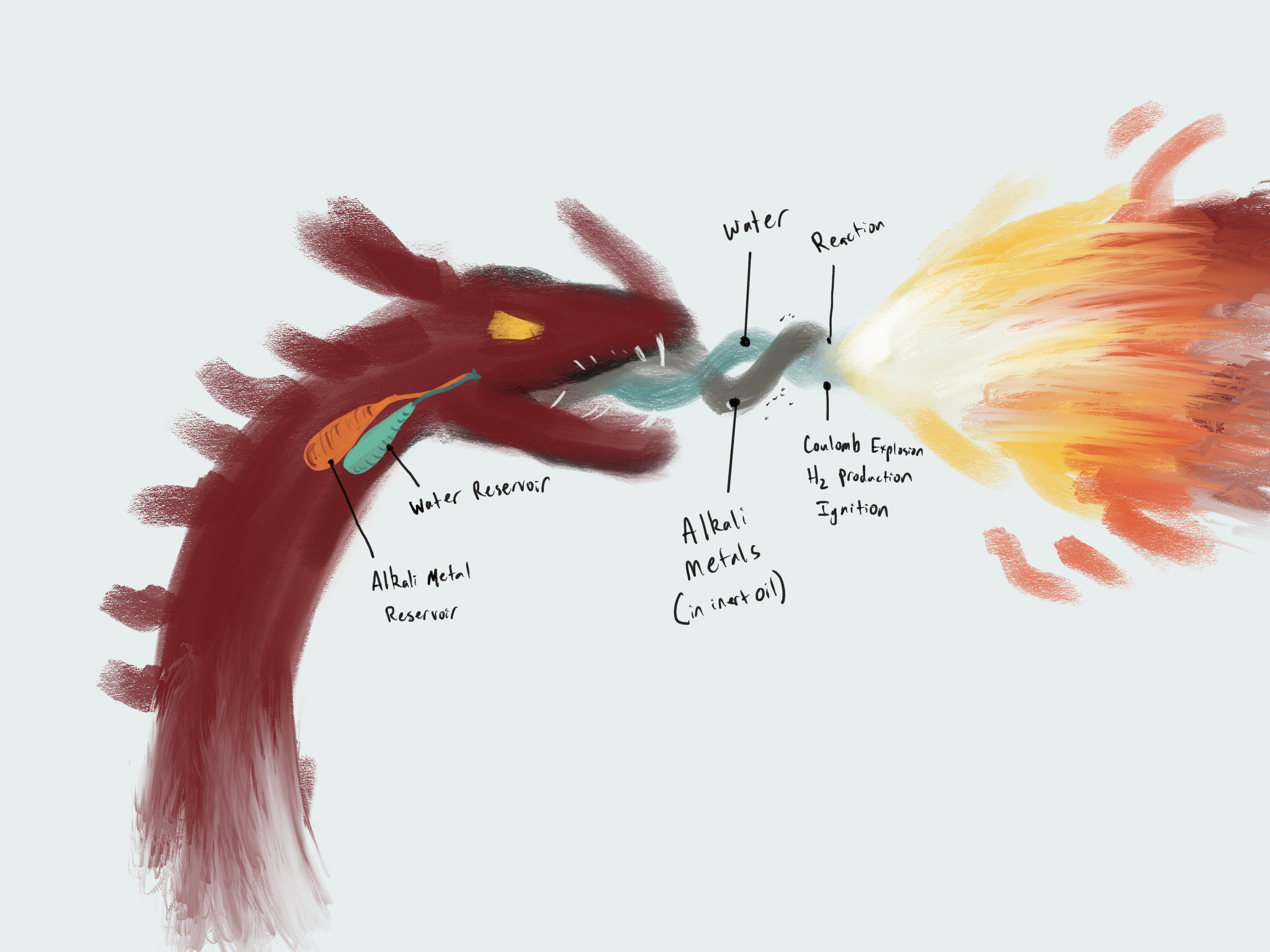
The dragon stores a cocktail or alloy of purified reactive metals in an internal pouch. Let’s call this this. Alkali metals have been isolated by the dragon’s metabolism (scientists are still figuring how that works) and the oil produced inside the dragon’s pouch is akin to mineral oil. This oil keeps the metals from reacting to the oxygen within the dragon (as a creature cannot truly be a low-oxygen environment). This way, our dragon doesn’t spontaneously combust from the internal metals.
The dragon opens their maw wide and spits out a wide, fan-like stream of the cocktail of reactive metals. From a secondary gland, they also unleash an impressive amount of specialized saliva. “Specialized” refers to the significantly low concentration of proteins and other organic compounds normally found in saliva. It’s effectively water, with slightly higher viscosity. Upon interaction with this water and the oxygen, all the reactive metals in the cocktail—lithium, sodium, potassium, magnesium, and so much more!—ignite!
It takes a moment for the metals to react, as each metal particle must lose the layer of mineral oil and interact with the water/oxygen. This reaction timeframe allows the stream to ignite far enough away from the dragon’s mouth and not burn the dragon.
Takeaways?
I haven’t even touched on the worldbuilding possibilities with this new biology!
Firstly, this would change the reasons driving dragon hunting, or even dragon rearing. If your dragon has a biological reservoir of ready-to-explode metals, you may have a subsection of dragon hunters who fight dragons to harvest that pouch. That metal could be used in a variety of different ways (my first thought was for warfare, like having blinding grenades you could throw at your enemies). Or, that metal could be used for organic chemistry much like it is in our modern world.
Maybe you want a more humane style of worldbuilding. You could have dragon tamers who raise domesticated dragons and have found ways to harvest the pure alkali metal that doesn’t harm the dragon (you know… put the dragon under anesthesia). This would mean that your dragon can only “breathe out” a stream of water, making your domestic dragon more of a personal “water hose” instead of “fire hazard.”
Wouldn’t it be wild if the fantasy culture’s version of fire fighters were these “de-metalled” dragons that hosed down flaming buildings? Talk about a role reversal.
Go write those fire—or water—breathing dragons, friends! Explore the possibilities!
If you’d like to support me and help me put out more cool blog posts like this, please remember to check out my Patreon.
